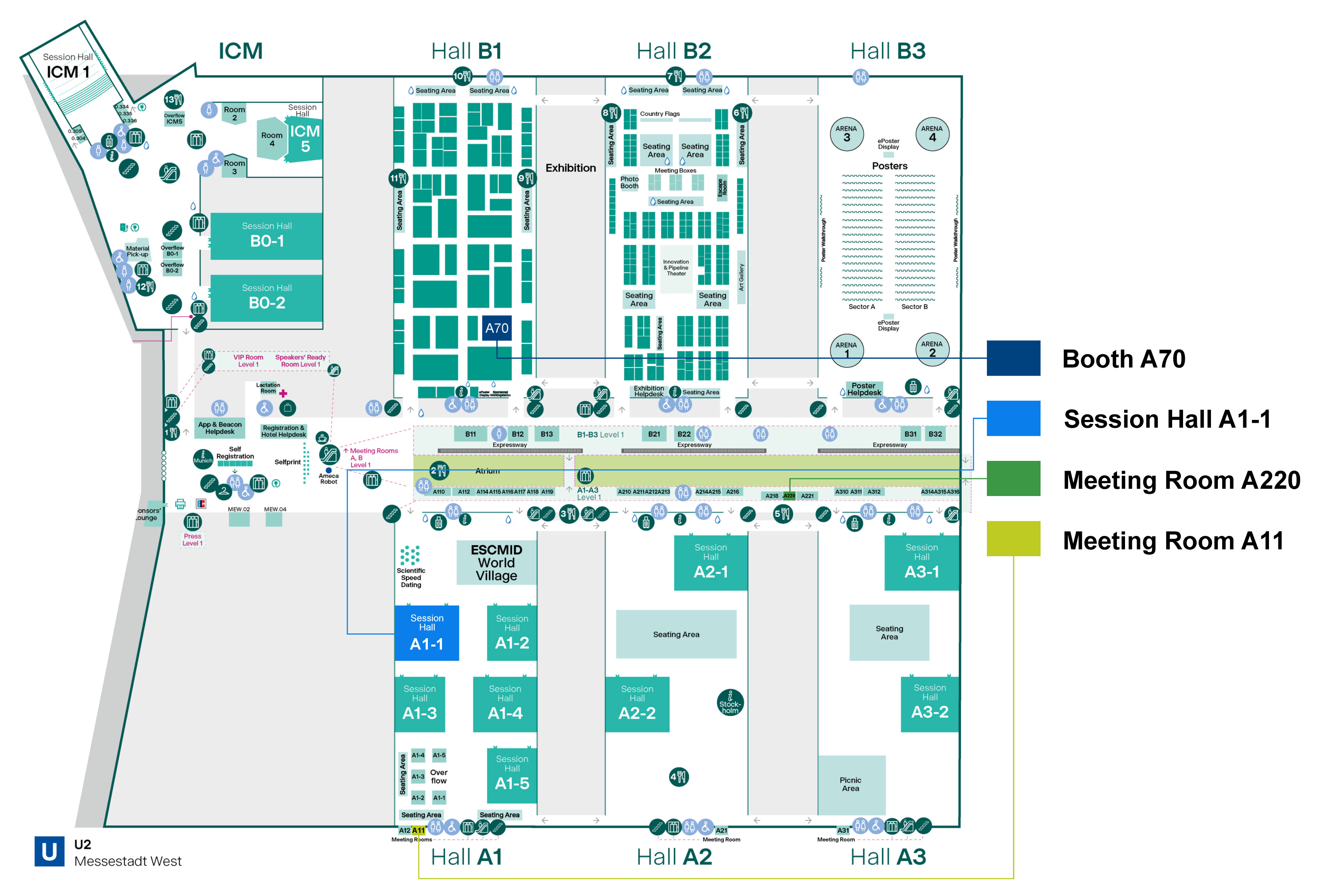
STRIDES* in AMR: Diagnostics Driving Impact
Sunday, April 19 | 16:15 – 17:45 | HALL A1-1
Tap into perspectives from leading experts on learnings from the OHE (Office of Health Economics) report: Taking STRIDES: The value of diagnostics against AMR.
Join us and gain:
- Clinical and pragmatic case studies
- Insights into implementation challenges & cost-effectiveness
- Diagnostic-driven improvements to stewardship programs, even in resource-constrained environments
Dr. Luke Moore - London, UK
Chelsea and Westminster Hospital NHS Foundation Trust
Dr. Maria Virginia Villegas - Bogota, Colombia
Universidad El Bosque
Dr. Erin McCreary - Pennsylvania, USA
University of Pittsburgh Medical Center
Pr. Dr. med. Mathias Pletz (Organiser-appointed chair) - Germany
University Hospital Jena, Institute for Infectious Diseases and Infection Control Jena
Dr. Gert Marais (ESCMID-appointed Chair) - South Africa
University of Cape Town, Medical Microbiology
Syndromic Testing Impact & the Evidence Behind it
Monday, April 20 | 12:15 – 13:15 | HALL A1-1
What impact could syndromic testing have for your standard clinical practice? Join us to explore guidelines, evidence and best practices with leading specialists.
Come discover:
- Practical & emerging diagnostic stewardship approaches
- Pragmatic insights and guidelines for syndromic testing adoption
- Real-world clinical applications & outcomes
Dr. Benjamin Berinson - Hamburg, Germany
Medical Center Hamburg-Eppendorf (UKE),
Pr. Ignacio Martin-Loeches - Dublin, Ireland
Trinity College Dublin Consultant, Intensive Care Medicine St James's Hospital
Dr. Kristie Johnson - Baltimore, Maryland, USA
University of Maryland School of Medicine,
Dr. Andrea Prinzi (Organiser-appointed chair) - Salt Lake City, USA
bioMérieux
Dr. Anna Both (ESCMID-appointed) - Hamburg, Germany
Medical Center Hamburg-Eppendorf (UKE), Cell Biology & Infection
PIONEERING DIAGNOSTICS
Visit us at BOOTH A70

ANTIMICROBIAL STEWARDSHIP
Advance antimicrobial stewardship with integrated diagnostics and data-driven solutions designed to detect, target, and treat drug-resistant infections. Optimize Diagnostics. Optimize Therapy. With our complete solutions for AMS.
LEADING MICROBIOLOGY
Improving antimicrobial use and sepsis outcomes with advanced ID & AST.

ADVANCING SYNDROMIC MOLECULAR
Empowering confident care with fast, comprehensive syndromic results.
CONNECTING DATA TO DECISIONS
Enhancing therapeutic decisions through data-driven insights.
BOOTH HIGHLIGHTS
VITEK® SOLUTIONS
Leverage a complete automated offer for identification and antimicrobial susceptibility testing that truly pushes back technology limits to innovate microbiology.

BIOFIRE® FILMARRAY® TORCH &
BIOFIRE® SPOTFIRE®
Take the guesswork out of syndromic PCR testing. Fast, actionable results in as little as ~15 mins to ~1 hr can equip clinicians with the information they need to provide pathogen-guided care and improve outcomes.

BIOMÉRIEUX
VISION SUITE
Power the shift from instrument-centric laboratories to data-driven infectious disease management—extending insights from the laboratory to clinicians and national surveillance. A comprehensive suite of solutions: MAESTRIA™, CLARION™, LUMED™, BIOFIRE® FIREWORKS®

BACT/ALERT® VIRTUO®
Get faster results with a fully-automated blood culture system designed to refine and redesign laboratory workflow, maximizing efficiency with the fastest clinically impactful results on the market.1,2

WATCHFIRE™ SOLUTION
The WATCHFIRE™ Solution, part of the BIOFIRE range, offers multiplex PCR testing in wastewater for key pathogens involved in disease outbreaks. Optimize wastewater surveillance to proactively manage potential public health risks by informing communities with real-time, on-site data, trending and analytics.
VETFIRE™ Equine Respiratory Panel
Syndromic approach to accurately detect and identify the pathogens most commonly associated with equine respiratory infections.
TUBERCULOSIS TESTING

AmPORE‑TB® RUO
AMR gene characterization for resistant strains with AmPORE‑TB® RUO.
Designed by Oxford Nanopore Technologies, manufactured by Oxford Nanopore Diagnostics - a fully owned subsidiary of Oxford Nanopore Technologies plc. Oxford Nanopore Technologies products are not intended for use for health assessment or to diagnose, treat, mitigate, cure, or prevent any disease or condition. Not available in the United States.
- 1. Destache et al. Diagn Microbiol Infect Dis. 2023 Dec;107(4):116085
- 2. Halperin at al. Microbiol Spectr. 2022 Oct 26;10(5):e0143622.
Product availability varies by country.
Please consult your bioMérieux representative.
VISIT US AT ESCMID GLOBAL 2026
Medical & Scientific Corner
BIOFIRE® Selection of Publications
ESCMID Global 2026 Special Edition
Evidence-based Diagnostics for Bloodstream Infection Management Selection of Publications
Edition 2026
VITEK® 2 Selection of Publications
Edition 2025
Antimicrobial Stewardship Educational Booklet
A practical guide to implementation in hospitals
Contact Us
Fill out the form for more information or to make an appointment to meet us at our ESCMID booth A70. We’re here to help!